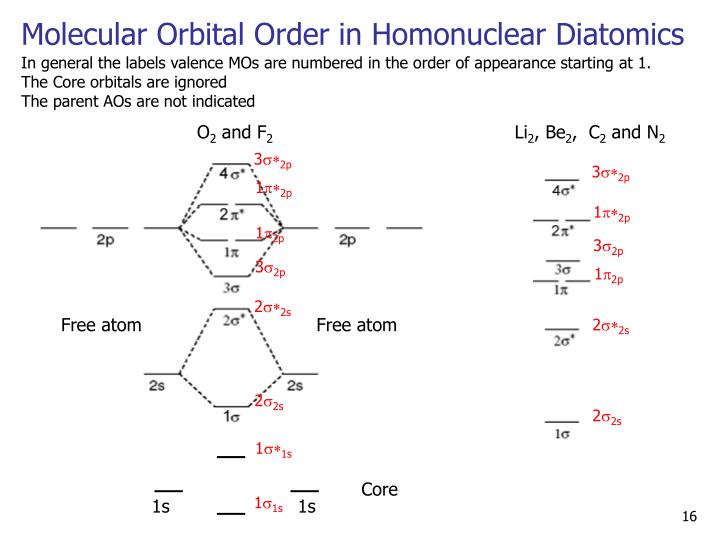
This means that the two electrons can have the same value of the three quantum numbers n, l, and ml, but the opposite spin quantum number. “Only two electrons may exist in the same orbital, and these electrons must have opposite spin,” according to the Pauli exclusion principle. This principle states that no two electrons in the same atom can have the same set of four quantum numbers. The exclusion principle, proposed by Austrian scientist Wolfgang Pauli, limits the number of electrons that can be occupied in certain orbitals (1926). The electrons fill each orbital in the following order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p, and so on. When two orbitals have the same (n+l) values, the orbital with the lower n value has the lower energy. The smaller the (n+l) number, the lower the orbital energy. The (n+l) rule can be used to predict the order in which the energy of orbitals grows, with the energy level of the orbital determined by the sum of the primary and azimuthal quantum numbers. This indicates that electrons only enter higher-energy orbitals once the lower-energy orbitals are completely occupied. The Aufbau Principle’s Key FeaturesĪccording to the Aufbau principle, electrons inhabit the lowest-energy orbitals first. The filling of electrons in an orbital during the creation of an electronic configuration is the subject of this principle. Aufbau is a German word that means “building up,” unlike many other chemical concepts. 
Noble gases are difficult to mix with other molecules because their orbital arrangements are so important. Furthermore, the most stable electron configuration has an energy state that is full. When all of an atom’s orbitals are filled, it becomes the most stable and, as a result, unreactive. The maximum number of electrons for the nth shell is 2n2, as established by a simple formula. 
The number of electrons in each shell is set. Electron configuration can be used to forecast an atom’s stability. The number of electrons in a molecule’s bonding and antibonding molecular orbitals can be calculated using the electronic configuration of the molecule or molecular ion. The dispersion of electrons in various molecular orbitals is referred to as a molecule’s electronic configuration. It’s critical that you know the difference between the two. The truth is that electrons do indeed reside in space-time regions known as orbitals.Īlthough the terms orbits and orbitals sound similar, they have very different meanings. The electrons may have been portrayed as revolving around the nucleus in a simplistic depiction of the atom. When a planet orbits the sun, it can be plotted on a precise route called an orbit. The Aufbau principle, which is based on Pauli’s exclusion principle, Hund’s rule of maximum multiplicity, and the relative energies of the orbitals, governs the filling of electrons into the orbitals of distinct atoms. The (n+l) rule is used to calculate the energy of an orbital. “The orbitals of the atom are filled in the sequence of their increasing energies in the ground state.” As a result, electrons first occupy the lowest energy orbitals available to them before progressing to higher energy orbitals.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
